Updated on 6/24/19
To avoid a citation for non-compliance with the June 2 Centers for Medicare & Medicaid Services (CMS) “requirement to reduce Legionella risk in healthcare facility water systems to prevent cases and outbreaks of Legionnaires’ disease (LD),” hospitals and nursing homes must “demonstrate measures to minimize the risk of LD.”
How the required outcome is accomplished CMS leaves largely up to each facility, simply referring to the water management program framework outlined in ANSI/ASHRAE Standard 188 and the CDC’s publication (toolkit), “Developing a Water Management Program to Reduce Legionella Growth & Spread in Buildings: A Practical Guide to Implementing Industry Standards.”
What a facility puts into that framework will determine success or failure with its water management program. For this article, let’s define success as more than just checking a box to avoid fines and citations. Real success is doing that and effectively minimizing health risk for the people in the facilities, reducing the organization’s legal risk, and protecting the organization’s brand and revenue–all without more money, time, personnel, water, or chemicals than necessary.
Based on a first-hand view of hundreds of facility water management programs since 2000, real success—for all facility types, not just healthcare—depends on these seven keys:
1. A Smart Hazard Analysis
Although a good water management plan (WMP) will include control measures to reduce the risk of many pathogens, Legionella is the best pathogen by which to define the scope and objective of a WMP because it is almost entirely waterborne, several commercial laboratories in the United States can test for it in water samples, and numerous scientific studies provide an abundance of data on which to base control measures and remediation methods.
If the stated objective of the WMP is to control Legionella, the hazard analysis amounts to determining which water systems present a significant risk of Legionella growth and transmission and an opportunity to reduce risk through proper management.
The determination is important. If systems that should be included are not, you could miss opportunities to prevent disease and will increase your legal risk. If systems that should not be included are, you will waste money on unnecessary control measures.
Common hazard analysis mistakes include these three:
- Determining whether to establish a WMP based on Legionella test results rather than types of water systems. Some water systems are inherently prone to Legionella which is why ASHRAE 188 requires a management program for certain water system types regardless of Legionella results. For hospitals and long-term care facilities participating in Medicare, CMS requires policies and procedures (control measures) for all building water systems that can harbor and spread Legionella and other opportunistic pathogens including plumbing (domestic water) systems and devices connected to them, whirlpool spas, humidifiers, decorative fountains, and cooling towers.
- Excluding domestic (plumbing) systems based on the criteria listed in ASHRAE 188 section 5.2. Many examples could be listed here: Small tank-type electric water heaters can be as or more prone to Legionella contamination than centralized domestic hot water systems. Domestic cold water systems—which in hot climates are actually not cold during several months of the year—must be properly managed because they supply hot water systems as well as ice machines, faucets, and showers. Moreover, they include the main feeds to the building, for which management is crucial, especially during construction projects or if one or more feeds is used only as a back-up. Also, Legionnaires’ disease has been linked to many buildings with fewer than 10 stories, another ASHRAE 188 risk factor.
- Inconsistency among properties. For example, if your organization owns 20 properties, you should be consistent in the control measures you implement for like-kind water systems and equipment, even if some properties are in a state with regulations and others are not. Some control measures may vary slightly based on the susceptibility of the building population to waterborne infections but, overall, equipment and system operation and maintenance should be consistent.
The hazard analysis is like a rudder. Getting it right is crucial to the rest of the WMP.
2. Smart Control Measures
Control measures are the most important part of a water management program (WMP) because what you do to your water is what reduces the risk of disease. Other parts of your WMP—flow diagrams, water systems descriptions, sampling—are important but in themselves do not reduce risk.
Answer these questions to find out if you are on the right track with your control measures:
- Does your WMP include control measures for all water systems (see #1 above, regarding the hazard analysis)?
- For each control measure, have you (per ASHRAE 188) listed a practical yet defensible performance limit, monitoring procedure for that limit, and corrective action if the limit is exceeded?
- Have you included measures for design and construction as well as operation and maintenance?
- Are all applicable measures included for each water system type? Many WMPs omit procedures that cost little or nothing but are crucial to reducing risk, simply because the facility or the company they hire to provide their WMP is unaware of them. Look at a WMP known to be comprehensive and see how yours measures up.
- Are your measures specific? Vague policies like “reduce stagnation” or “avoid dead-legs” are ineffective. Each control measure should be written such that a maintenance person could go do it (e.g., “Open the valve on the North Tower backflow preventer bypass for 30 seconds once a month.”).
- Are your measures evidence-based? Doing what is unnecessary or ineffective wastes time and money and sometimes water, energy, and chemicals as well.
3. Smart Training
Your personnel must know how to perform the control measures well. Also knowing why is even better.
Find the combination of training that works best for your organization. Classroom training has benefits of focused attention on the subject but is costly and time consuming for the material covered. Walk-through training is effective but restricted to a small number of people and limited content. Online courses require some self-discipline but are a low-cost way to present a lot of content in a relatively short period of time, on each student’s schedule.
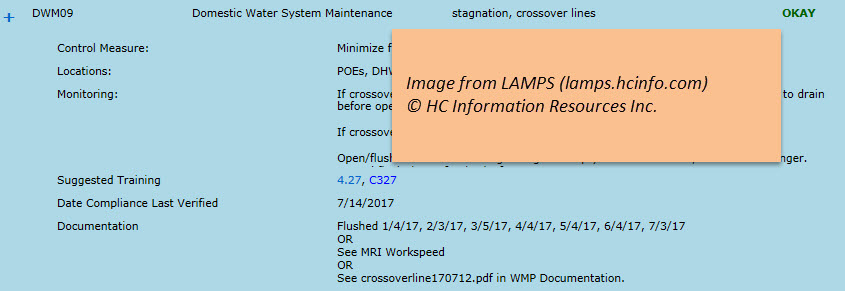
Training built into a WMP is especially effective because it provides the material needed at exactly the right time, to the right people. For example, most of the control measures in LAMPS WMPs include a “Suggested Training” link that, when clicked, takes the user immediately to a pertinent online course, short article, illustration, or diagram (see the link “4.27” in the image below).
4. Smart Execution
How you manage the development and implementation of your WMP will affect your cost and risk.
Facilities that execute well seem to have certain habits in common, one of which is striking the right balance between in-house personnel and outside services. If you attempt to write a WMP on your own from scratch, you could, like so many others, fool yourself into thinking you are saving money only to find you wasted countless hours on a WMP that is vague, incomplete, and indefensible.
Most make the opposite mistake—attempting to rely entirely on a vendor to develop and implement their WMP. That does not work, in part because many control measures will be performed better and much less expensively by your own maintenance personnel. To adequately reduce risk and control costs, facilities need to involve both in-house personnel and vendors.
Two facilities with the same WMP content can have quite different outcomes if one executes their WMP better than the other. In a September 2016 live webcast, I interviewed the facilities director, infection control director, and risk management director at UCSF Medical Center about the execution of their WMP because they have done it so extraordinarily well. Their success could be for several reasons but two stood out in the interview: They work hard at communication (e.g., scheduling regular conference calls with all parties to discuss the progress of implementing control measures) and they plan and monitor repair and construction projects thoroughly and vigilantly (e.g., to ensure minimal stagnation and biofilm disruption). (LAMPS users can watch the recording of the webcast by clicking on “Videos” in the training content.)
5. Smart Documentation
Documentation provides evidence of what you did and when. It also leads to the more consistent performance of the control measures.
You can document in various ways—inspection logs, vendor reports, work order software. The most practical way will vary with control measures. Be sure to have a hub—one location that shows how and where the control measure is documented. In LAMPS, for example, the control measure documentation field is the hub (see image above). In that box can be the documentation itself (e.g., “flushed 1/4/17, 2/3/17, and 3/5/17”) or reference to a log, work order software, or vendor report.
6. Smart Validation
Validation procedures measure the effectiveness of a WMP in accomplishing its main objective, i.e., to reduce the hazard (e.g., Legionella).
Testing water samples for Legionella provides the most reliable feedback on Legionella control. Chlorine measurements, temperate readings, and total bacteria counts can provide good information for water system management but in most water systems will not reliably indicate Legionella. However, for Legionella tests to provide good validation of a WMP, the sampling, laboratory analysis, and interpretation of results must be performed properly. Success depends on these six factors:
- Collect the optimum number of samples. Too many samples result in overspending. Too few provide data insufficient for decision making—a total waste of money and time.
- Plan samples strategically. Hot versus cold water. Pre- versus post-flush. The right locations.
- Collect samples properly. It is easy to fill a bottle with water but the details are crucial.
- Record data to maximize useful information. You get more for your money if you collect the right data.
- Use a proven laboratory. Don’t skimp here. Use the best you can find. At a minimum, ensure it has CDC ELITE Certification for Legionella testing.
- Interpret results from multiple angles, not just one. If you interpret results based on a simple grid of Legionella concentrations or positivity (percentage of plumbing system samples in which Legionella is found), you will miss opportunities to prevent disease, spend too much money on remediation, harm the environment unnecessarily, or all the above.
7. Smart Remediation
Too many facilities and consultants take an all or nothing approach to domestic water remediation, responding to Legionella test results by either doing nothing or disinfecting the entire system. Rarely is either extreme the best action.
Don’t be like the facilities that have responded inadequately or too late to test results—deciding instead to just retest, hoping the results get better. And don’t waste money on ineffective changes, like facilities that have replaced all domestic water piping at the advice of an engineering firm—and still failed to fix a Legionella problem that could have been solved for $30,000.
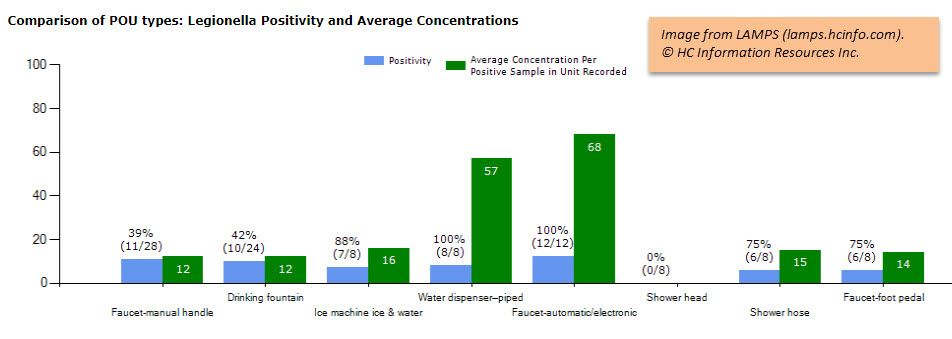
Thorough and properly interpreted analytics is the key to smart remediation. Instead of making decisions on a simple Legionella threshold, compare results for hot versus cold water, buildings, and outlet types. The facility with the results shown in the image below should consider replacing shower hoses more often, since they had higher positivity than showerheads over several sampling rounds, and either discontinuing the piped-in water dispensers or replacing them with a different model.
Using analytics to pinpoint the root of problems allows you to remediate with a rifle instead of a shotgun—solving problems faster, more effectively, and at a much lower cost.
Facilities that implement water management programs based on a smart hazard analysis and with smart control measures, training, execution, documentation, validation, and remediation will reduce health and legal risks without overspending or wastefulness.
Update June 24, 2019: In July 2018, the CMS updated its requirement (QSO-17-30), clarifying expectations and specifically noting that environmental testing for pathogens is at the discretion of the facility. ASHRAE has also updated Standard 188, making it more regulation-ready.
What else have you found to be important for water management program success? You can leave a comment below.


Great webinar and list of Key Success Factors!
Completely agree on the importance of using a lab with demonstrated competence in Legionella testing. However, although CDC ELITE is a well know program for identifying laboratories for outbreak testing, the BEST determination of competence for Legionella and all other types of testing is through nationally recognized accreditation such as: The National Environmental Laboratory Accreditation Program (NELAP), ISO 17025 or American Industrial Hygiene Association (AIHA) EMLAP program.
Accredited environmental laboratories are required to participate in at least the following in order to maintain their Accreditation, including for Legionella testing, if it is on their Scope of Accreditation. To be clear, the CDC ELITE Program is not an accreditation program.
Laboratory Accreditation requires that a laboratory must (at minimum) have:
• A written Quality System Manual that outlines all aspects of laboratory management, testing and training and includes:
o Human Resources
o Document control
o Standard Operation Procedures (SOP) for each method on the Accreditation Scope of work
o Defined Corrective and Preventative Actions
o Chain of Custody for samples
o Initial and On-Going Technical capability assessments for all technicians for each test method they perform
• Regular 3rd party audits
• Laboratory must pass Annual, or Bi-annual Proficiency Test (PT) samples from an accredited PT provider
• Quality Control checks are used with each Batch run of samples
• Data reports are reviewed and certified by a Quality Assurance Manager
Thanks for providing the information, Kristin. I appreciate it.
Hello Kristin,
You are absolutely right about ISO17025 accreditation and the like.
I had been in charge of laboratories for over 15 years, and I cannot have a different opinion.
In any case, sampling, shipping and technician rotations are important factors in making the wrong analysis.
I think we now have enough opportunity to do a test on site, and increase the frequency to have better follow-up due to the low prices of such a solution.
Is not it ?
Enjoyed the webinar – thanks for putting this on and sharing the information. Please stay in touch!
Thanks Christoph!
Great info.
One more element of “smart validation” and meeting the CMS requirement is focusing on getting the best possible quantification of the primary pathogenic species of Legionella, L. pneumophila. Legionella pneumohila has been the cause of 99% CDC outbreaks of Legionnaires Disease and 96% of all culture confirmed cases reported in Europe in data through 2015 so it’s essential to get accurate, repeatable and reproducible data on that specific species. The amount of other Legionella species is not relevant for routine monitoring and ensuring you’re taking the right water management steps.
Thanks for the additional information, Kristin. I agree it’s essential to get accurate results for Legionella pneumophila. I disagree, however, that finding other Legionella species in building water systems is irrelevant to the management of those systems.
This was a great webinar. The 7 keys to success will be helpful to building owners and their staff when developing a water management program, especially now that healthcare facilities must develop a program and perform “risk assessments” of their water systems for legionella and other opportunistic pathogens. Special attention to how the records of the plan will be documented and updated accordingly is needed. The building owner is ultimately responsible for the safety of their staff and occupants so they need to “own” their plan and ensure it is being followed as designed.
Just to clarify, NSF International is a non-profit, non-governmental organization that is developing NSF/ANSI 444 with members of organizations such as ASHRAE and the CDC in order to prevent disease and injury from legionella and other opportunistic pathogens, as well as chemical and physical hazards.
Thank you for the clarifications, Andrew
great webinar….one question…what exposure does a water treatment company have if the customer does not implement a Legionella Water Management program…We have the biocide treatment in accordance to “best practice”
Thanks Mike. Although I won’t give legal advice, my guess is that your responsibility will be limited to the tasks you are obligated to perform per your agreement with the facility. Typically, nearly all the responsibility for implementing a WMP lies with the facility.
I took your two day course over 20 years ago and you sparked a continued interest in the subject. One comment, in # 2 Smart Control Measures you recommend bleeding the back flow preventer by pass. A back flow preventer should never have a by pass. The bleeding can be accomplished by opening the back flow preventer test cocks.
Hi Dick. Many of the bypasses around back flow preventers that I’ve seen have back flow preventers on them. Some of them don’t, though. Thanks for pointing out that that is bad practice.
Found the webinar very informative
Thanks Dale!
All man-made water systems can be a breeding ground for Legionella and the conditions on board ship provide the perfect environment for the bacteria to thrive. You should be paying particular attention to these areas:
– Any part of your water system that has a temperature between 20–50 °C.
– Any outlet that creates and disperses water droplets. This includes showers, sink taps, air conditioning units, fire hoses, and washing equipment.
– Anywhere that water is stored or re-circulated.
– Places where there are deposits containing nutrients that allow bacteria to grow such as rust, sludge, scale, organic matter, and biofilms.
There are a few simple measures that you can perform to reduce the risks from Legionella.